 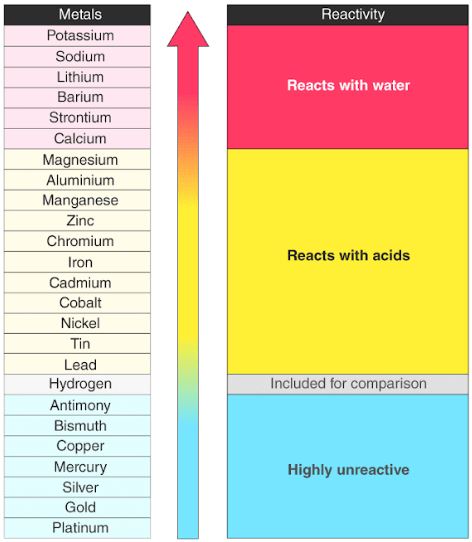
As you go up a group the amount of shells an atom has decreases which pulls the valence electrons in the outermost shell closer to the nucleus and increases an atom’s ability to attract more electrons To The protons in its nucleus. 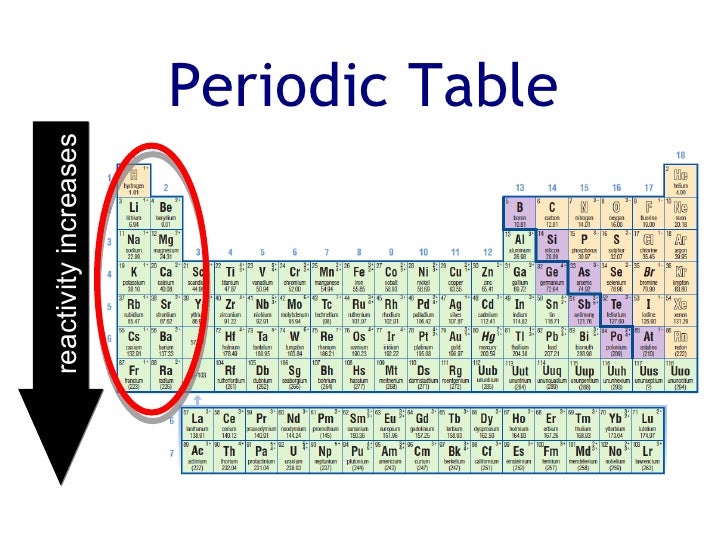
Francium is the most reactive metal! reactivity of non-metals: As you go left to right within a period, the atom is getting a closer to obtaining a full valence shell, so the more readily it will react to receive those electrons. As you go down a group, the valence electrons move farther away from the nucleus (more shells) which make them easier to be given/taken away. As the electrons move closer to the nucleus, It will require more energy to remove them (High ionization energy) and It will be easier for the atom to attract more electrons (High electronegativity).Ĥ REACTIVITY OF Metals: As you go from right to left within a period, the amount of valence electrons a Metal has to give away decreases, so It will become more readily available to react. 
As the number of Protons and valence electrons Increase, The attraction increases which pulls the outermost shell closer to the nucleus (Opposites Attract). Thus, it will require less energy (lower Ionization energy) to remove an electron from the outermost shell and It will also be more difficult To attract electrons (low electronegativity)ģ Within a period, elements have the same number Of Shells, But Increase in the number Valence electrons as you travel across a period. As the number Of shells increase, the valence electrons are moved closer away from the nucleus. Phenomenon: Three periodic trends (atomic radius, ionization energy, and electronegativity) help chemists determine an element’s reactivity in creating compounds.Ģ Within a Group, elements have the same number of valence electrons but as you go down a group, the number of shells increase. Unlike the other elements in period 3, it is not fully oxidised to its highest oxidation state of +6 – it only reaches an oxidation state of +4.Ī catalyst and a high temperature is required to reach +6 and form SO₃.Presentation on theme: "PERIODIC TABLE TRENDS & REACTIVITY"- Presentation transcript: Sulfur reacts with oxygen to form sulfur dioxide. White phosphorus will react spontaneously with oxygen to produce a white smoke of phosphorus pentoxide. Red phosphorus must be heated before it reacts with oxygen. These are different physical forms of the same element. Silicon will react with oxygen quite slowly to produce silicon dioxide. It will burn brightly to produce a white powder of aluminium oxide. The reactions gives a bright white flame and leaves a white powder of magnesium oxide.Īluminium will react quite slowly with oxygen unless it is in a powdered form. (a) How does the chemical reactivity of alkali metals vary on going down in group 1 of the periodic table (b) How does the chemical reactivity of the halogens. So, we can generally think of reactivity in terms of electronegativity and/or electron affinity. Less reactive elements tend to share electrons. More reactive elements tend to gain or lose electrons. Magnesium ribbon burns readily in oxygen to produce magnesium oxide. Reactivity is an element's ability to ionize (gain or lose electrons) or share electrons. Sodium will react directly with oxygen and burns brightly to give a characteristic bright yellow flame. When magnesium is heated with steam, the following, very exothermic reaction occurs: Reactions with OxygenĪll Period 3 elements can form oxides when they react with oxygen. The reaction has a much higher activation energy Reactivity of Period 3 Elements This is why magnesium is less reactive than sodium. This results in a greater electrostatic attraction holding the magnesium lattice together. Magnesium has a smaller atomic radius and higher charge compared with sodium. Reactivity of Period 3 Elements Why is Magnesium less reactive than Sodium? This is because magnesium is less soluble in water in water than sodium. The resulting solution of this reaction is a weak alkaline – not as strongly alkaline as the one produced by the reaction between sodium and water. It is a very slow reaction at room temperature. Magnesium reacts less violently and less rapidly than sodium. The resulting solution is a very strongly alkaline solution of sodium hydroxide, this has a pH of about 13-14. The sodium will fizz and float on the surface of the water as it reacts. Sodium reacts with water in a rapid and violent reaction. The only metal elements in period 3 that will react with cold water are sodium and magnesium. Sodium (Group 1) has 1 outer electron in the third outer shell. The group number tells you the number of electrons in the outermost shell – e.g. The period number tells you the number of shells – e.g. Period 3 elements are in the third row of the periodic table. Properties of Period 3 Elements - Reactivity of Period 3 Elements (A-Level Chemistry) Period 3 Elements PE - Use the periodic table as a model to predict the relative properties of elements based on the patterns of electrons in the outermost energy level of. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
